Abstract
Fiber alignment in the extracellular matrix provides critical topographical cues for tissue regeneration, yet the underlying epigenetic mechanisms remain poorly understood. Here, we developed 3D-printable hydrogel microfiber inks to systematically investigate how aligned topography drives muscle regeneration through epigenetic priming. Gelatin-norbornene (GelNB) microfibers (approximately 300 μm in diameter) were fabricated via ultraviolet crosslinking, fragmented, and jammed to create shear-thinning inks for extrusion-based 3D printing. Aligned microfiber scaffolds induced alignment of C2C12 murine skeletal myoblasts with increased nuclear aspect ratios and chromatin decondensation. An assay for transposase-accessible chromatin using sequencing revealed rapid chromatin remodeling within 24 h, with enrichment of master myogenic regulators and mechanosensitive transcription factors at newly accessible sites. RNA-seq confirmed widespread differential gene expression, demonstrating that physical alignment drives chromatin accessibility changes that enable transcriptional activation of muscle differentiation programs. In vivo validation using a volumetric muscle loss model revealed that compared with the perpendicular alignment of control groups, the groups with scaffolds with horizontally aligned fibers achieved superior muscle regeneration, with enhanced tissue recovery and reduced fibrosis. Epigenetic changes observed in vitro were confirmed in vivo, validating the mechanistic link between topographical cues and chromatin remodeling during tissue regeneration. These findings establish that 3D-printed aligned microfiber topography systematically controls epigenetic mechanisms to guide muscle regeneration, providing a powerful platform for the development of regenerative fibrous biomaterials.
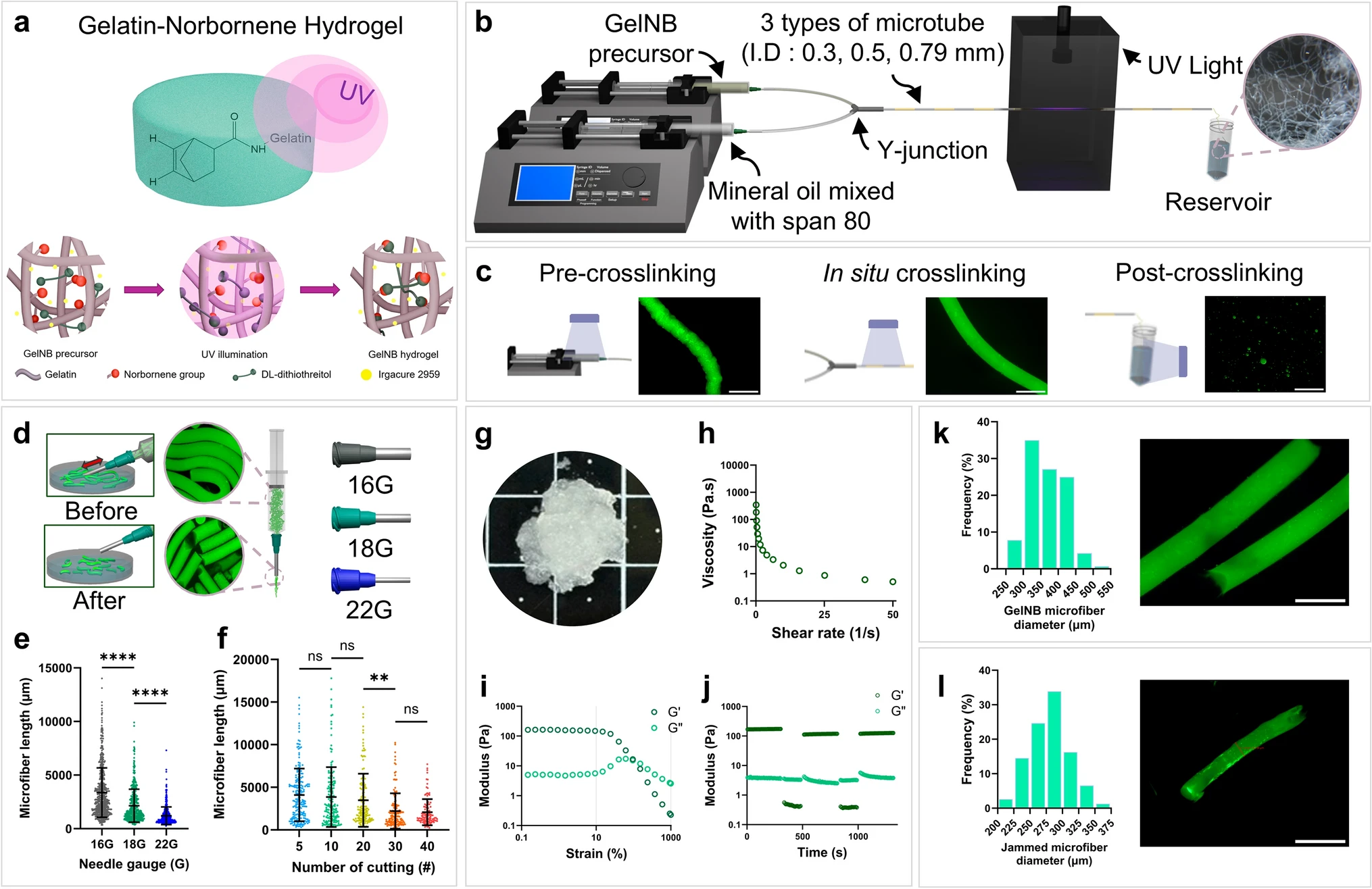
Fabrication and analysis of GelNB microfiber inks
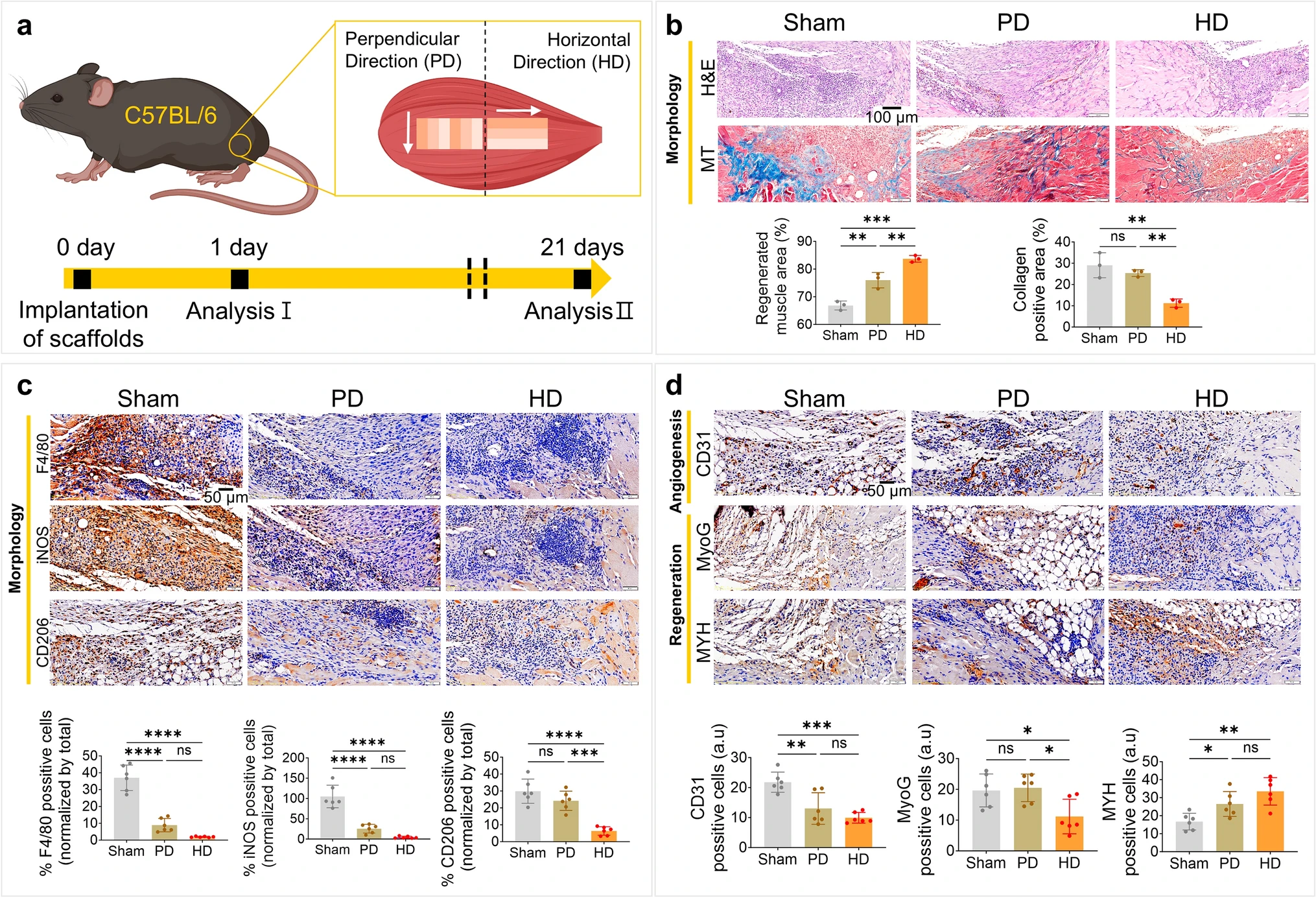
In vivo performance of aligned scaffolds